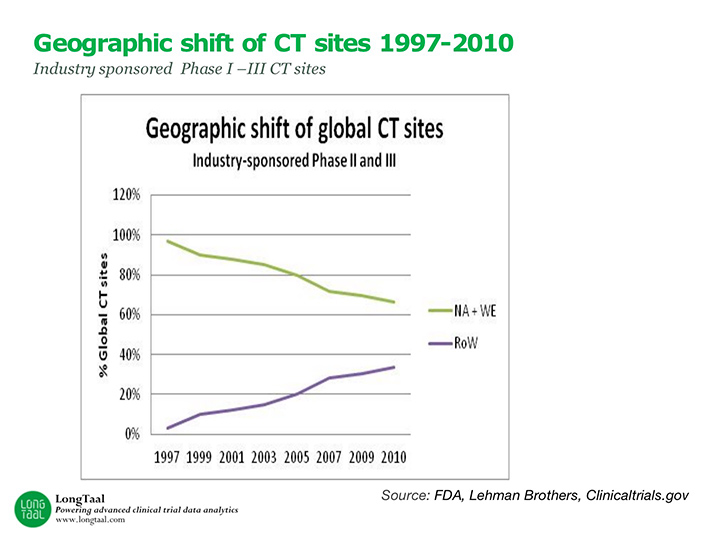
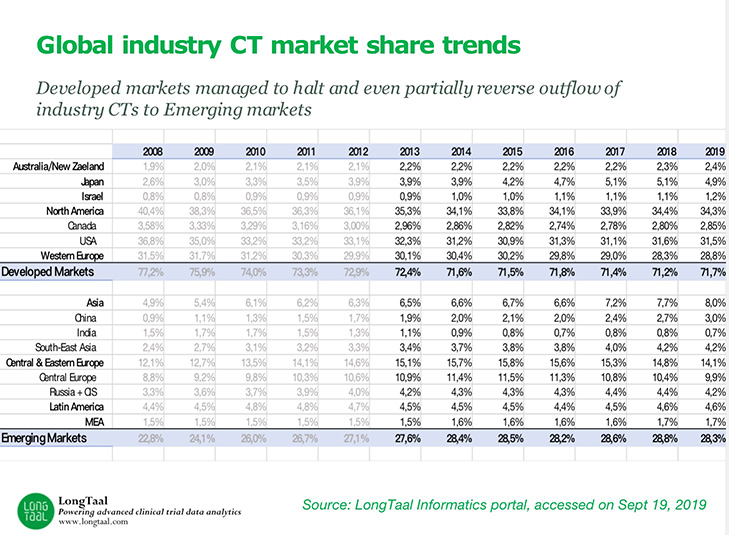
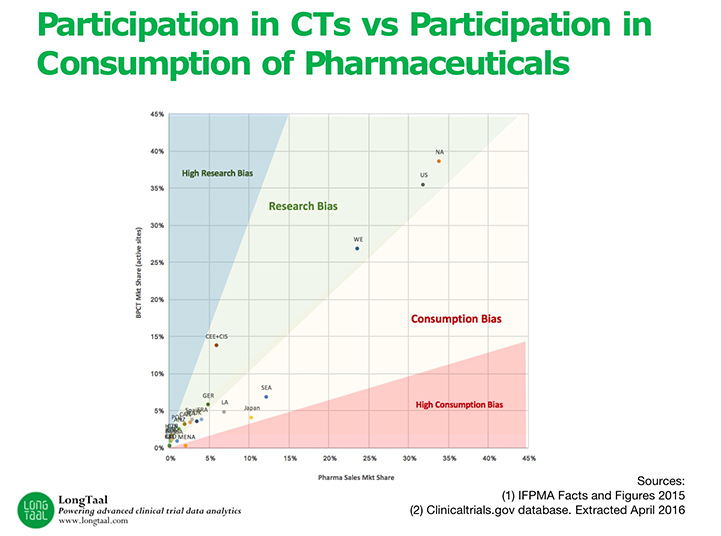
Episode Four: Evaluating Geographical Clinical Trial Trends with Dr. Vladimir Misik
Guests

Shunee Yee
Shunee Yee is the President and CEO of CSOFT International and CSOFT Health Sciences, overseeing operations across 3 continents. In her 25+ years of industry experience, Ms. Yee has been featured in numerous publications, including the Economist, Fortune Magazine, Forbes Asia, and IDG journals. She is also a Fortune selected Top 10 Most Powerful Women Entrepreneur in 2012 and was identified by CNN Money as one of technology’s 36 most powerful disrupters. In June 2016, Ms. Yee helped launch the Shenzhen 100 research report, utilizing qualitative indicators to support global market strategies. Her many contributions in the sphere of education include the Johns Hopkins-Nanjing US-China Center, Michelle Obama’s Let Girls Learn initiative, and her work on the board of trustees at Dexter Southfield School. Additionally, Ms. Yee is a member of the Joslin Diabetes Center’s A Taste of Ginger committee, supporting the Asian American Diabetes Initiative (AADI).

Dr. Vladimir Misik
Dr. Misik is the Partner and Founder of LongTaal, Partner and Co-founder of VIARES Academy, as well as a board member at SanaClis. With 30 years of biomedical R&D experience, Dr. Misik has held a range of previous roles within the industry and in academia, working as a VP and Head of Global Centralized Operations Center, Global Clinical Operations Leadership Team Member and Regional Head of CEE and MENA at Quintiles/IQVIA. Dr. Misik has published extensively on various aspects of globalization of industry clinical trials, contributed to oncology research at the National Cancer Institute in Bethesda in Maryland, as well as Cardiovascular Research at the George Washington University, School of Medicine. Dr. Misik is an Editorial Board Member at Applied Clinical Research, Clinical Trials and Regulatory Affairs as well as a member of the DIA Global Core Committee for Clinical Research.